|
Liquicop
is a liquid copper formulation with no crystalline nature. As a
result of this, Liquicop does not cause any wear on your spray
equipment, is easy to measure and does not leave your tractor
and driver covered in blue powder.
Why
is liquid better than
crystalline copper?
Crystalline copper (copper
oxychloride, copper hydroxide) is not very soluble, and at best
forms a suspension of small crystals. These crystals are then
pumped through your impeller, filters and nozzles and cause
excessive wear. The droplets land on the leaf and start drying
down. As this happens, the suspended micro- crystals act as seed
crystals for the small amount of soluble copper to re- crystalise
onto. This results in relatively large crystals settling loosely
on the leaf surface. The same can be said for the other “liquid”
coppers available on the Australian market which are simply
crystalline copper in suspension.
Liquicop,
on the other hand, is never a crystal - in fact it is manufactured
from a solid piece of copper which is brought into the solution by
electrolysis. Once it is mixed (no pre- mixing required) and
pumped smoothly through your spray- rig, the droplet starts drying
down on the leaf. There are no seed crystals so the copper starts
to crystalise onto the leaf surface (trichomes and cuticle).
Texture of various leaf surfaces provide anchorage for Liquicop e.
g. A: Trichomes (hairs) on Kalanchoe leaf, B: Onion cuticle
These
crystals are now the same as crystaline copper, but are firmly
attached to the leaf, and coat the leaf surface, rather than as
lumps of copper on the leaf. Which would you rather use to keep
bugs out of your house - insect screen or burglar bars? The finer
the gaps in your defence, the less chance of a successful
infiltration.
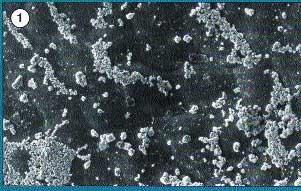
Copper hydroxide on a leaf
surface. Note the size of crystals and how they concentrate
and leave gaps on the leaf (800 times magnification).
|

Liquicop on the leaf surface.
Note fine crystal formation and even distribution over the
surface (800 times magnification). |
The
Liquicop crystals are also much smaller (i. e. their surface area to
mass ratio is higher) and have more surface area for re- dissolving
whenever there is irrigation or dew - high risk periods for fungal
and bacterial infection. The better adhesion of crystals to the leaf
does however extend withholding periods.
Why is pH
important with copper sprays?
The optimum pH for all copper sprays is pH 6- 7.
With crystalline coppers this is less critical as so little copper
is in the solution that phytotoxicity is minimal, but so is the
effectiveness. With Liquicop which is 100% soluble, pH is more
important. As you decrease the pH, the risk for burning increases -
as you increase the pH the formation of copper oxide increase.
Copper oxide crystalises on the leaf but is almost completely
insoluble, leaving you no residual protection.
Mixing
instructions
Two factors are important:
1. Correct pH before adding any other chemicals.
2. Add Liquicop last to the mix. A fully tested
compatibility list is available. Preferably check the pH prior
to adding Liquicop to complicated cocktails.
How does copper
control
disease?
Liquicop interferes with enzyme
function and protein synthesis in pathogens.
Other benefits:
1. Lower concentration of copper in
Liquicop reduces the possibility of copper toxicity in the leaf
or in the soil.
2. Easily dosed by volume.
3. No unsightly blue residues on
crops due to more even distribution.
4. Safe for use in high
concentration aerial applications as long as adequate coverage
is achieved.
5. Liquicop is environmentally
compatible, and has no effect on pollinators.
6. Liquicop’s formulation should
not cause flower drop in sensitive crops, but should preferably
not be applied at peak flowering periods.
By: Steve Ansermino
|